 Jerry Avorn, M.D. Co-Founder & Special Adviser, NaRCAD Tags: Evidence Based Medicine, Jerry Avorn Following the astonishing debut of AI applications like ChatGPT a year ago, “knowledge workers” (that’s us) have been forced to ponder how much of what we do could be replaced by a very smart set of computer programs. Such applications can already pass medical licensing exams better than many graduates and have gotten remarkably good at reading X-rays and pathology specimens. How soon will AI systems become adept at reviewing the clinical literature and preparing concise, user-friendly summaries, complete with prescribing recommendations? Not yet, but likely before long.  Try it yourself at home: log onto OpenAI.com (it’s free) and ask ChatGPT for advice about medications for diabetes or hypertension or HIV or anything else. Just be careful about its “hallucinations” – the fact that sometimes AI just makes up wrong stuff. (I prefer the term “confabulation,” also used to describe this well-known phenomenon.) That can be whimsical if you’re a N.Y. Times reporter and ChatGPT advises you to leave your spouse, and it can be very problematic if you’re a lawyer who relies on case law that ChatGPT simply fabricated. (Both actually happened.) But it can be lethal if it involves incorrect clinical recommendations. Yet that said, AI is getting smarter every day. If programmed well in the coming years, large language models like ChatGPT or its growing number of competitors could eventually also learn how to gauge prescribers’ current knowledge, attitudes, and practices, and then ask just the right questions to find out why they’re doing what they’re doing, what their concerns are, and what it would take to get them to change.  Once things mature a bit further, will large health care systems interested in academic detailing and in cost-cutting simply replace humans with AI-AD-bots? After all, they could work 18-hour days, don’t need health care benefits, and can disseminate any message their employer wants. It will be easy replace a recommendation like “SGLT-2 inhibitors in diabetes can reduce cardiovascular and renal disease as well as lower glucose” with: “SGLT-2 inhibitors are extremely expensive and increase our drug budget. Use metformin or sulfonylureas whenever possible.  So if we have a few years to prove that actual people still have a vital role to play in helping practitioners make better decisions, what can we do?
Those are values that endure and can distinguish our work from a sophisticated set of algorithms. Best of all, they can’t be changed if whoever is in charge overwrites a few lines of code to maximize some other agenda, or if the algorithms just make stuff up. Biography.
Jerry Avorn, MD, Co-Founder & Special Adviser, NaRCAD Dr. Avorn is Professor of Medicine at Harvard Medical School and Chief Emeritus of the Division of Pharmacoepidemiology and Pharmacoeconomics (DoPE) at Brigham & Women's Hospital. A general internist, geriatrician, and drug epidemiologist, he pioneered the concept of academic detailing and is recognized internationally as a leading expert on this topic and on optimal medication use, particularly in the elderly. Read More. Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD Tags: Stigma, Evidence-Based Medicine, Health Disparities, Conference Missed our event? Check out the AD Summit videos and materials on our Summit Hub.  We’re fresh off of the excitement of hosting our Academic Detailing Virtual Summit, “A Deeper Understanding of Our Impact on Patient Care.” In prioritizing patient-informed care more than ever before, we explored patient narratives through live interviews, workshops, and special panels, all within a virtual space. Innovations included AD for criminal justice involvement, care delivery redesign for veterans, affirming care for transgender and nonbinary people, and patient-informed communication on sex positivity in HIV prevention. Take a peek at some of the highlights from our event below! AD Fireside Chat: A New Spin on a “Keynote Address” We opened our AD Summit with a real-time interview featuring the originator and co-founder of NaRCAD, Dr. Jerry Avorn, and the National Director of the Veterans Affairs Pharmacy Benefits Management Academic Detailing Service, Melissa Christopher. Audience members were excited to throw ideas around and ask questions about:
Program Development Workshops: Attendees Led the Charge! Our revamped course catalog of workshops invited attendees to be in the director’s seat, as well as behind the scenes as co-creators in small groups. Participants created resources that we’ve published on our website and social media channels, sharing creativity and expertise with the larger AD community. Our workshops covered a wide range of topics including:
 The Pleasure Project The Pleasure Project Special Panel: Understanding Critical Care Needed for Formerly Incarcerated Patients The outstanding team from New York City Department of Health and Mental Hygiene (NYCDOHMH) shared their groundbreaking detailing campaign, “Public Health Detailing for Criminal Justice Involvement”, with an audience that was hungry for innovation around inclusivity. The NYCDOHMH team shared NYC clinicians’ understanding of formerly incarcerated patients’ care, including clinicians who met the campaign with stigma, and those who were grateful to see such a campaign being implemented. Best Practices Spotlight: Prioritizing the Patient Experience For the first time in NaRCAD’s history, we highlighted leaders in the field who’ve been prioritizing the patient experience. The San Francisco team created space for conversation and discussion about gender-affirming care and ways to encourage safe clinical environments for transgender and nonbinary individuals through language, storytelling, and community outreach. We also heard from the Arizona team about the importance of pleasure being part of a patient’s sexual health history and the role of a detailer in supporting these conversations between clinicians and patients.  Inclusivity Roundtable: Real-time Script Creation We wrapped up our AD Summit with a roundtable session where attendees co-created a scripting resource to empower detailers to combat stigma during visits. We asked attendees to come up with responses to the stigmatizing comments below. A five-page resource was created in 60 minutes! 1. “I can’t believe patients keep coming back without having lost weight. They’re clearly not trying hard enough, and not making healthy food choices.” 2. “I don’t want those patients at my practice. They’re so difficult to handle and are really just looking for another opioid prescription. Treatment won’t work for them.” 3. “I’m so tired of keeping up with all these different pronouns. You’re either a man or a woman. It gets in the way of providing care.” 4. “I don’t need to use an assessment tool. I can always tell when someone’s at high risk of contracting HIV.” We want to continue these conversations, hear about your team’s innovations, and share resources in person this fall at our annual conference in Boston, MA. We hope to see you there! -The NaRCAD Team A special thank you to all of our AD Summit attendees and presenters as well as our partners at the Agency for Healthcare Research and Quality. For more information on our presenters, you can view the AD Summit Program Book. Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation! By Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD An interview with Adriane Apicelli, MSW, Project Manager, Harm Reduction Projects, University of New Hampshire, College of Health and Human Services. Tags: Harm Reduction, Detailing Visits, Evidence-Based Medicine  Anna: Hi, Adriane. Harm reduction is deeply meaningful to many individuals – can you share why harm reduction is meaningful to you? Adriane: The core principles of harm reduction, such as self-determination, mutualism, and self-advocacy, resonate with my personal and professional values. I firmly believe that individuals are the experts in their own lives, and it’s essential that people have access to the necessary resources, materials, and support systems to ensure their safety and well-being. Harm reduction also offers a powerful framework that facilitates a shift within communities that moves away from moralization and stigmatization of individuals who use drugs. It’s easy to blame individuals for their behavior, but it’s far more challenging to critically reflect on how systems and decisions put people in those circumstances in the first place. Anna: By embracing harm reduction principles, people within our communities can foster empathy and understanding for those facing challenges with substance use. Everyone deserves the same dignity, respect, and access to necessary resources. Can you tell me about the harm reduction detailing project you’ve been working on in collaboration with the National Association of County and City Health Officials (NACCHO)? Adriane: We’re currently focused on detailing elected officials in the State of New Hampshire, including city counselors, mayors, and senators. The opinions and decisions of elected officials shape the harm reduction service landscape – they make decisions that either enable or constrain available resources. Anna: Recognizing the influential role of elected officials is crucial when working on public health initiatives, including harm reduction. What is the overall goal of your current detailing project?  Adriane: We want to empower elected officials to make informed decisions that increase capacity of harm reduction services based on the needs of their communities. We’re currently encouraging elected officials to establish direct connections with individuals who use drugs so they can integrate the expertise from those with lived and living experience into decision-making processes regarding laws, policies, and resource allocation. Anna: Actively listening to those with lived and living experience helps to better understand how to support specific populations. How did your team decide to focus on encouraging elected officials to directly connect with people who use drugs? Adriane: During the development of our detailing project, we consulted with local syringe service program (SSP) participants and asked them how they think elected officials can better understand substance use and harm reduction. The overwhelming response was for direct communication between individuals who use drugs and elected officials, or having elected officials spend the day with them to understand their experiences firsthand. We’re trying to figure out how we can facilitate these approaches to ensure the safety and ethical treatment of SSP participants. We’ve also been exploring the possibility of forming advisory committees to incorporate the perspectives of individuals who use drugs in the decision-making process in a safe and supportive environment.  Anna: Advisory committees certainly help to ensure that voices and perspectives are heard and valued. Let’s transition to thinking about all of your detailing work to date – what makes you most proud to be a harm reduction detailer? Adriane: The people I detail. We shouldn’t underestimate how hard it is to change our minds, our attitudes, or our behaviors. It takes so much humility and effort to receive and integrate new information, especially when it counters your social values and beliefs. It’s an honor to work through that learning process with those that I detail. Anna: Do you have a specific example of that learning process that you can share? Adriane: I detailed someone who was initially hesitant to publicly announce that she prescribes buprenorphine because she was worried how that information would affect her patient panel. We ended up having a conversation about substance use stigma and its implications. We discussed that openly sharing that she prescribes buprenorphine serves as a powerful signal to patients, assuring them that she provides a safe environment to seek treatment. It also sends a message to other clinicians about the importance of prescribing this medication to patients who need it.  Anna: Having those types of honest conversations with people you detail is imperative to changing behaviors and reducing stigma at the individual and community level. Is there anything else you’d like to add before we wrap up today? Adriane: Remember that it’s much easier to build harm reduction capacity in collaboration with others. Last year, I collaborated with individuals from the public health department, a local hospital, the New Hampshire Harm Reduction Coalition (NHHRC), and a community volunteer to address a concern raised by a business owner regarding improperly discarded sharps on their property. We formed an informal work group and created a proposal aimed at piloting an anonymous syringe disposal project, installing two disposal units in the community. The disposal units were proposed to be on city property, so we needed buy-in from City Council to be able to do this. We recognized the power of engaging elected officials and presented our proposal to the City Council. Our proposal received unanimous support and it’s currently being piloted in the community. Collaborative advocacy and engagement with members of the community and elected officials can bring about positive change and enhance the health of all. Anna: We often have more power than we think when we collaborate with others who have similar goals. Detailing is an effective approach for encouraging collaboration and connection with experts in the community, including experts with lived and living experience. Thanks for joining us today, Adriane! We look forward to continuing to hear about your inspiring harm reduction work in New Hampshire. Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. Since 2020, Adriane Apicelli has served as the Project Manager and primary academic detailer of the University of New Hampshire (UNH), Department of Nursing’s Harm Reduction Education and Technical Assistance (HRETA) project. She holds a Master of Social Work (MSW) from Boston College, where she also earned a Certificate in Management. In addition to her role with the HRETA project, Adriane serves as a nonprofit strategic planning consultant and has previously served as an adjunct professor for the Department of Social Work at UNH. By Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD An interview with Rocko Cook, Public Health Detailer and Public Health Detailing Program Manager, Detailing Improved Sexual Health (DISH), Arizona State University. Tags: Detailing Visits, Evidence-Based Medicine, HIV/AIDS  Anna: Hi, Rocko – thanks for joining DETAILS today! HIV prevention work is critical and often deeply meaningful to those working in the field. Can you tell me why this work speaks to you? Rocko: I found out I was positive for HIV and syphilis in 2002. I encountered firsthand the stigma and shame associated with these diagnoses. It was devastating, but it’s made me feel even more connected to the work I’m currently involved in. I’ve worked in various roles in the field of HIV prevention and care since 1994, just two years before medication was widely available. In the 90s, there was little support for people who tested positive for HIV from a care perspective, financial perspective, and housing perspective. We now have the medications and tools to prevent, treat, and support people with HIV. Despite this, there’s an urgent need to continue educating clinicians, staff, case managers, community health workers, and other people who are on the frontlines about these tools and resources. Academic detailing helps to close that gap and gives me the opportunity to live my passion of educating others about HIV.  Anna: Thank you for your openness in sharing your personal and professional experiences. Your passion for HIV prevention and care is evident. What’s the number one thing you want clinicians and staff in your community to do differently when it comes to HIV? Rocko: I’d love for them to change the culture of the entire clinic space and create a more welcoming environment for patients. There needs to be a focus on consistent communication coming from every professional a patient meets with when receiving care. All clinicians and staff need to be able to communicate with patients in a sex-positive way and in a way that connects with patients’ specific experiences, identities, and needs. They need to be comfortable communicating about sexual behavior, testing, and PrEP. It’s difficult to have these conversations. I've been doing this for a long time and I’m not perfect at it either, but once we practice and start getting comfortable with ourselves, then it gets a lot easier to be comfortable with patients. Anna: Modeling this type of communication during a detailing visit is key. It can help clinicians and staff feel more comfortable having the same conversations with patients. Can you share a story from the field about a positive response or reaction from someone you detailed? Rocko: There was an agency we worked with that hired a new physician, testers, and medical assistants for their mobile medical and HIV testing unit. They had never worked with this patient population, so our team did several trainings and 1:1 detailing sessions where we role played conversations with patients. We needed to bring them up to speed on how to have gender inclusive conversations and communicate with sex positivity. We had a lot of fun together. The team ended up going to Phoenix Pride to do a big testing event. We were delighted with their success in providing testing to the community and creating a welcoming and safe environment for people interested in being tested.  Anna: That’s a large event for the mobile unit team to tackle, while also succeeding in creating a safe space for all! Let’s talk a little bit more about the impact on patients. Can you share any data on the impact of your detailing work? Rocko: We have anecdotal evidence that folks are benefiting from our services. My colleague and I are closely involved in the gay community and people often tell us about their care experiences. We’re in an enviable position because we know a lot of people and hear things in passing. It helps us do a better job targeting our services; we can work directly with clinics that we’ve heard would benefit from detailing. I also recently connected with someone of trans experience who was tested at Phoenix Pride. They’ve had poor encounters in the past where clinicians and staff assumed the body parts they have. They shared that they had a positive experience with the mobile unit and felt comfortable throughout the visit. Being able to see our impact firsthand has been really motivating for me; it makes my heart sing. Anna: It’s rare to be so closely connected to the community that’s being impacted by your detailing work. It’s clearly been beneficial for your detailing efforts and getting your program up and running. Let’s wrap up with a final question - what has made you most proud of this project so far?  Rocko: I’m so proud of the way our team has come together and engaged with partners across the state. We’ve been able to leverage partnerships and community relationships to enhance program development, implementation, and dissemination. I’m also proud of our creativity in choosing our program’s name, DISH AZ (Detailing Improved Sexual Health). We send out a Weekly Special with a buffet of options on new evidence and information related to HIV prevention and care. We’re creating an active and robust network of professionals, while using food as our motivator! Anna: That’s an innovative way to keep your network engaged! We’re looking forward to hearing about your program as your team continues to expand its network and positively impact more people in the community. Thanks for chatting with us today and sharing your experiences, Rocko! Your passion for this work is palpable. Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. Rocko Cook serves as the Program Manager for DISH-AZ (Detailing for Improved Sexual Health in Arizona), a program of the Office of Evaluation and Partner Contracts for the Southwest Interdisciplinary Research Center (SIRC) at Arizona State University in partnership with Arizona Department of Health and Human Services. Rocko began working in the field of HIV in 1994 and is a community leader with over 15 years of experience implementing prevention and care programs in Arizona, Ohio, and Kentucky. In addition to his duties as a program manager, Rocko has served as a public speaker, presenter at local and national conferences, and as a consultant and leader for HIV community planning groups. Rocko has been living with HIV since 2002 and is passionate about improving sexual health for all communities. By Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD An interview with Meghan Breckling, PharmD, BCACP, Ambulatory Care Pharmacist and Academic Detailer, University of Arkansas for Medical Sciences and Arkansas Department of Health. Tags: Detailing Visits, Opioid Safety, Harm Reduction, Evidence-Based Medicine 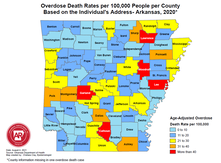 Overdose Deaths Rates per 100,000 people per County, Arkansas 2020 Overdose Deaths Rates per 100,000 people per County, Arkansas 2020 Anna: Hi, Meghan. Thanks for joining me on DETAILS today! Your team has done extensive work on pain management detailing, and you recently completed a pilot project on harm reduction in collaboration with the National Association of County and City Health Officials (NACCHO). Can you tell me a little more about this project? Meghan: Thanks for having me! We decided to target rural counties in Arkansas that have both high drug overdose deaths and naloxone administration rates. We previously created broad pain management materials for our other opioid safety detailing projects; this project took those materials to the next level. We looked at how we could better support clinicians in caring for their patients with substance use disorder (SUD) through a harm reduction lens. We provided clinicians with screening tools to help identify patients with mental health conditions and SUD to determine who could benefit from additional services. We even created a local resource guide for clinicians to easily connect patients to community services. The clinicians found that these accessible tools helped them have open conversations with patients.  Anna: I can imagine having something tangible to give to patients makes clinicians feel more equipped to have these conversations. What other resources were you able to share with clinicians? Meghan: We encouraged clinicians to utilize a new, free mental health resource called AR ConnectNow. This program provides immediate virtual care to all Arkansans dealing with mental health and substance use disorders. Clinicians were grateful for AR ConnectNow because mental health services are scarce in rural Arkansas; they’ve been sharing it with their patients frequently. Anna: You must have been proud to be part of a project that had such an impact on both patients and clinicians. How did the harm reduction lens inform your detailing visits for this project compared to your prior pain management-focused visits? Meghan: Many visits centered on communication with patients. Communication and empathy are two huge pieces to consider with this topic. We spent a lot of time asking clinicians about the conversations they have with patients and the types of questions they ask about substance use. We really wanted to understand what was going well and where there were gaps that we could help fill with resources and support. We also focused on naloxone prescribing and administration. We gave out free naloxone kits to all clinicians that they could either keep in the clinic or give to a patient who was having trouble accessing it. Clinicians were open to the idea of prescribing naloxone to patients who were at risk of overdose and open to keeping kits in their clinic in the event of an overdose. Our team had a lot of clinicians say during follow up visits that they felt more comfortable prescribing naloxone and were prescribing it more to patients and family members.  Anna: It’s impressive how you were able to clearly shift your focus from opioid prescribing to harm reduction and prioritize the relationship between the clinician and patient. Did you receive any pushback from clinicians on harm reduction? Meghan: Clinicians understood the need for harm reduction services but were more inclined to refer patients out rather than providing services within their clinics. For example, we found that a lot of clinicians were resistant to prescribing Medications for Opioid Use Disorder (MOUD), either because they were uncomfortable with the steps to do so, or they were told by leadership that they should not prescribe MOUD at their practice. It can sometimes take an hour or more for patients in rural areas to access specialty services that offer MOUD. We’re looking at future projects where we can utilize pharmacists to increase MOUD prescribing in partnership with primary care providers. For instance, a primary care clinician could diagnose SUD and prescribe MOUD, while a pharmacist could monitor the patient throughout treatment. It would take a lot of burden off the clinicians and could possibly make them less resistant to prescribing it.  Anna: Using pharmacists as an integral part of the care team is an excellent idea – you’ll have to let us know if you receive additional funding for this work! Let’s wrap up with a final question. If another program decided to do a detailing project on harm reduction, what advice would you give them before they went out into the field? Meghan: You need to take a step back and remember that there isn’t going to be instant behavior change among clinicians. For a topic this complex, it’s critical to have follow-up visits and continue to be a resource and support for clinicians. Also, be understanding of clinicians and their experiences. They’re dealing with a lot and it’s not easy to change things all at once. Building a relationship and getting a clinician to commit to just one key message is a huge win. Want to learn more? Read about the harm reduction key messages used for this project and the development of those messages on our previous blog post. Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. Dr. Meghan Breckling is an Ambulatory Care Pharmacy Specialist at the University of Arkansas for Medical Sciences (UAMS) and is a trained Academic Detailer through the National Resource Center for Academic Detailing (NARCAD) within the Center for Health Services Research (CHSR) at UAMS’ Psychiatric Research Institute (PRI). She previously completed a PGY1 Pharmacy Residency and PGY2 Ambulatory Care Residency at the Central Arkansas Veterans Healthcare System (CAVHS). Currently, she is a part of a multidisciplinary academic detailing team comprised of a pharmacist, physician and physical therapist that provide evidence-based solutions, tools and support for chronic pain management to primary care providers across the state of Arkansas. By Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD An interview with Shuchin Shukla, MD, MPH, Faculty Physician, Mountain Area Health Education Center (MAHEC), NaRCAD Training Facilitator Tags: Harm Reduction, Detailing Visits, Evidence-Based Medicine, Opioid Safety  Anna: Welcome to the DETAILS blog, Shuchin! You wear many hats - you’re an addiction medicine physician, an academic detailer, and an academic detailing trainer. Tell us how you got started with academic detailing. Shuchin: I had an interest in marginalized populations and did my residency in the Bronx in New York City. I was a clinician in HIV care for several years before moving with my family to Western North Carolina. Soon after we moved, I began working at our Area Health Education Center (AHEC) and it was evident that addiction was the primary public health and clinical issue that was causing the most harm in my community. Fast forward a few years and one of my colleagues received a Centers for Disease Control and Prevention (CDC) grant and asked if I could attend a NaRCAD training to learn more about AD. The medical board and one of our pharmacists at the Department of Public Health were very interested in using AD for overdose prevention. We started with a pilot where we detailed 10 clinicians and slowly built our program. We now have multiple AD grants we’re working on, including one on adverse childhood experiences (ACEs) and one on harm reduction.  Anna: We often tell programs to start with a small pilot before growing their programs so that they can identify what went well and where there are opportunities for improvement, especially on new detailing topics like ACEs (e.g., key message adoption, clinician response, etc.). You do a lot of education around substance use disorder and mentioned that your team received a harm reduction grant for AD – what does harm reduction mean to you? Shuchin: The goal is, simply enough, to reduce the level of harm that a person may be facing. Harm reduction means having no expectation of a person's behavior and accepting the reality of what people live and do without judgement. It’s about being open with patients so that they’re more likely to come back for a visit where you can continue to have a conversation with them about getting a little bit healthier. There’s evidence to support harm reduction. The research shows that providing harm reduction services, whether it's naloxone or syringe exchange, reduces harm, but also decreases substance use and helps people engage in substance use care and treatment. Anna: Do you see harm reduction being used with other topics beyond substance use disorder?  Shuchin: There are tons of examples of harm reduction that are built into everything we do. Seatbelts, masks, fire escapes, smoke detectors, vaccines, and the FDA regulatory agency are all forms of harm reduction. As a society, we’ve never looked at substance use through this lens because using drugs is so stigmatized. Anna: I imagine it’s difficult to have detailing visits with clinicians because of the type of stigma associated with it, such as thinking that it’s some sort of moral failing. How have clinicians responded to detailing visits on harm reduction? Shuchin: Most of the teaching about harm reduction is unlearning all the inaccurate information we've been taught. We're taught if you use drugs, you're a bad person and you should be penalized. I remember watching the show Cops growing up and there was always a person of color laid out on a car resisting arrest. Law enforcement would pull out a bag of cocaine from the car and say they’ve saved the community. None of this is right, but I saw that on TV as a middle school kid. It’s easy to generate a lot of negative energy about substance use disorder and substances in general from these shows, and clinicians are part of that thinking too. Asking clinicians to talk to their patients about harm reduction is a lot different than asking them to check their Prescription Drug Monitoring Programs (PDMPs) to ensure that patients aren’t receiving multiple prescriptions for controlled substances. Having a conversation with a patient takes empathy and thoughtfulness, whereas checking a PDMP does not. We’ve found that clinicians who have been the most resistant to harm reduction are those who have family members with substance use disorders. They are often angry, and rightfully so.  Anna: It’s imperative to be empathetic during detailing visits, especially on a topic that affects so many people. Let’s explore harm reduction from a different angle. How do your patients respond when you bring up harm reduction during your clinic visits? Shuchin: These are certainly challenging conversations to have, so you need to start off by letting patients know that they aren’t going to get in trouble for sharing this information and you need to acknowledge the trauma and stigma that surrounds substance use. Patients seem grateful that I approach conversations in a straightforward way that doesn’t stigmatize their use of drugs. I’ve never had a patient be offended or confused about why I was talking to them about harm reduction. Their eyes usually widen when I ask them things like how they use their drugs, how they cook their drugs, or where they get their drugs from. They often say, “I’ve never had a doctor like you.” Anna: You must spend a lot of time building trusting relationships with patients so that you can have these conversations. Shuchin: I do. It also helps that the organization I work for, our county commissioners, and our sheriff are all on board with harm reduction. There’s a lot of focus on Naloxone distribution among members of our community, such as law enforcement, first responders, and other clinicians. Our clinic prescribes a lot of medications for opioid use disorder, specifically buprenorphine, which is also a form of harm reduction. We have peer support specialists who meet patients where they’re at and start the conversation about harm reduction with them before they even have their first visit with me.  Anna: It’s definitely critical to have a community that supports the way you practice, as well your program’s AD messaging. Can you share a final tip for other detailers who are working on harm reduction? Shuchin: Harm reduction is an emotional topic for a lot of people, especially folks who are in frequent contact with people who use drugs, like emergency room clinicians or people with lived experience in their families. With this topic, paying attention to the emotions of the clinician you're detailing and acknowledging those emotions before jumping into your key messages is much more important than any other topic I’ve worked on. Be patient and empathetic – every visit counts toward making a change. Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. Shuchin Shukla, MD, MPH, was born and raised in New Orleans, Louisiana. He completed medical school and public health school at Tulane University and completed a residency in family medicine at Montefiore Medical Center in the Bronx, New York. He worked in the South Bronx for 5 years following residency, providing primary care for adults and children, as well as for adults living with HIV. He also served as medical director for Montefiore Project INSPIRE, a primary care-based Hepatitis C treatment program. He then moved with his family to Asheville, North Carolina, where he currently serves at Mountain Area Health Education Center (MAHEC) as faculty physician and Clinical Director of Health Integration. He is an associate clinical professor of medicine in the Department of Family Medicine at the School of Medicine, University of North Carolina in Chapel Hill, and is a Diplomate of the American Board of Preventive Medicine, Board-Certified in Addiction Medicine. Additionally, he is a Robert Wood Johnson Clinical Scholar. He leads on various initiatives and projects around addiction, HIV, Hepatitis C, homelessness, and the criminal justice system. His main experience as a detailer has been focused on improving evidence-based provider interventions related to opioids, pain, and addiction. By Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD An interview with Jennifer Carefoot, BSc, Pharm, Academic Detailing Pharmacist, BC Provincial Academic Detailing (PAD) Service. Tags: Conference, COVID-19, Detailing Visits, Evidence-Based Medicine  Anna: Hi, Jen! Thanks for joining us on DETAILS today. Earlier this year, your team developed a detailing campaign in one week for the new COVID-19 treatment, Paxlovid, and presented on your work at our NaRCAD conference. I’m excited to learn more about this campaign. Before we jump in, can you give us some background about your organization? Jen: The British Columbia Provincial Academic Detailing Service (BC PAD) launched in 2008 and provides academic detailing to the province of British Columbia. We have 12 full-time pharmacist detailers for the entire province. The program is publicly funded by the BC Ministry of Health and our topics are chosen based on their relevance to primary care clinicians. Here are some quick facts about our program:
 Anna: Your team connects with many clinicians covering a vast geographical area! Can you walk us through the one-week development of the Paxlovid campaign that you presented on at our conference? Jen: Paxlovid was approved in Canada on January 17, 2022. The Province of BC quickly received doses and urgently needed educational sessions for clinicians in the province for safe and appropriate use of this somewhat complicated medication. The PAD team had the expertise in delivering pharmacotherapy and education through detailing visits; we worked with the Ministry of Health and the COVID Therapeutics Committee (CTC) to quickly develop materials. We were out in the field detailing within a week! Anna: That’s an impressive turnaround time to get your team prepared and out in the field! Were clinicians aware of Paxlovid when your team started detailing on the topic?  Jen: Paxlovid was getting a lot of press in the media at the time- it was the first oral treatment readily available for COVID-19. Clinicians knew they would have to know about this medication since their patients would be asking about it. Anna: New information was coming out every day in the media, which isn’t very common for other detailing topics. How was your team able to stay up to date on the emerging evidence on Paxlovid and continue to keep clinicians up to date? Jen: Our team was able to stay up to date through the excellent communication between our PAD director, Terryn Naumann, the CTC, and the Ministry of Health. We would receive updates via email or video conference daily. We also frequently met internally to share questions, successes, and challenges that we were experiencing in the field. To keep clinicians up to date, we chose to not provide them with the PowerPoint presentation from the detailing visits because the information and evidence was constantly changing. We’ve created 24 versions to date! Instead, we emailed a one-page resource to clinicians after our visits that included links to online resources that were updated in real time.  Anna: That’s one of the benefits of sharing resources virtually – the information can be updated and accessed quickly. If you were told you had to create another campaign in one week, what would you do differently? Jen: That’s a tough question! Would we have liked more detailers? Yes. Do I wish there were 24 more hours in my day? Yes. Would we have liked more clinical trial data? Yes. These are all things that are outside of our control. We did the best we could with the evidence and resources we had. I don’t know if we would do it any other way if we had to do it again under the same circumstances. Anna: What you did worked. Do you have any evaluation data from clinicians that you can share? Jen: Our formal evaluation showed:
 Anna: I’m not surprised by these results! Your team has shown impressive data from myriad detailing campaigns. Before we wrap up, can you tell us how your partnerships and team helped make this campaign successful? Jen: It’s important to partner with others who have expertise in the therapeutic area you’re detailing on. You don’t need to reinvent the wheel with evidence. Pull from data and evidence that other AD programs or reliable organizations have previously put together and be open to the idea that evidence changes. Also, remember that it really does take a team! Everyone brought something critical and unique to the table. Our administration team was instrumental in getting our visits booked and our director was transparent with our team throughout the process. Our team was excited, enthusiastic, and proud to have the opportunity to provide this education to clinicians in our province. Anna: Having a team that is open to a challenge and eager to stay up to date on evidence is so important. It’s not realistic for programs to have to create a campaign in a week, but if they ever find themselves in that situation, we know we can all turn to you and your team for support! Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. Jen is an academic detailer for the BC Provincial Academic Service since 2016 serving beautiful South Vancouver Island. She is passionate about translating evidence to practice, reducing polypharmacy and shared decision making. In her spare time, she enjoys running, gardening, travelling, and spending time with her family outdoors at the lake. Honest Conversations: Supporting Clinicians in Linking Patients to Harm Reduction Services11/14/2022
By Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD Tags: Primary Care, Opioid Safety, Evidence Based Medicine, Harm Reduction  Our team at NaRCAD has been working on an exciting new project developing harm reduction key messages for primary care clinicians in collaboration with the National Association of County and City Health Officials (NACCHO), Centers for Disease Control and Prevention (CDC), and consultants from Boston Medical Center. The Substance Abuse and Mental Health Services Administration (SAMHSA) defines harm reduction as an approach that aims to prevent overdose and infectious disease transmission, improve physical and mental health, and offer options for accessing treatment and other health care services for people who use drugs. Various harm reduction approaches have been proven to prevent overdose and death, injury, infectious disease transmission, and substance misuse. For instance, there is nearly 30 years of research that has shown that syringe services programs decrease transmission of viral hepatitis, HIV, and other infections. There are several other harm reduction approaches beyond syringe service programs, including:
It’s critical that academic detailers continue to encourage primary care clinicians to discuss harm reduction with their patients and link them to services within their community. Academic detailers have the ability to empower clinicians to have difficult conversations with patients to reduce infections, overdose, and death. Our team developed the following key messages to support primary care clinicians in caring for patients who would benefit from harm reduction. These key messages are currently being piloted across the United States in a project funded by NACCHO.  Harm Reduction: Key Messages to Improve Outcomes for People Who Use Drugs 1. Assess factors that may contribute to risk of Opioid Use Disorder (OUD) for patients who use opioids. 2. Identify opportunities to reduce risk of harm using a patient-centered approach. 3. Offer Medications for Opioid Use Disorder (MOUD) to patients identified as having OUD. 4. Connect patients with community harm reduction services and other services that meet identified needs. These evidence-based key messages can help clinicians provide support to their patients and build strong and trusting relationships with those who need it most. Building trust between clinicians and patients allows patients to feel heard and be open to seeking additional treatment, ultimately leading to improved health outcomes. Our team is looking forward to continuing to explore harm reduction and updating our key messages based on the results of the pilot through NACCHO. If your program is interested in collaborating with our team on future harm reduction work, or any other clinical topic, please reach out to us at [email protected]. Want to learn more? Stay tuned to learn about the results of the pilot and how clinicians responded to these key messages in the field. You can also join our discussion forum to interact with peers who are working on harm reduction!  Jerry Avorn, M.D. Professor of Medicine, Harvard Medical School Co-Founder & Special Adviser, NaRCAD Tags: Evidence Based Medicine, Jerry Avorn Inside the world of academic detailing, we’re all convinced of the clinical power of information transmission when done well, and of the harm that can result if it’s done badly or not at all. But outside our mighty little community, many are skeptical about whether pushing out accurate evidence really has such enormous effects. A silver lining of Covid-19 as it recedes a bit into the rearview mirror is that the experience can help us put some of those questions to rest once and for all. Many lives were changed by the effective, accurate transmission of medical reality about the virus, how to prevent its terrible consequences, and how best to treat people who are infected. Thousands of doctors and millions of patients were offered and acted on accurate information about covid, and the point was nailed down by its counter-factual: the millions of patients (and yes, some doctors and health care professionals as well) who received and acted on bad information, with tragic consequences. It was the same virus, more or less, the same vaccines, the same effective and ineffective treatments: sort of a controlled study in which effective transmission of evidence-based facts was lifesaving, and its opposite was often lethal.  Covid was just the most dramatic and recent example of issues that all of us interested in academic detailing have been grappling with for years. It was like a student coming to class one day to confront a surprise pop quiz that will determine three-quarters of the grade. Those who had been diligent all semester could deal with it handily, while those who hadn't been paying enough attention are astonished and devastated. It’s the same for the transmission of clear information about most of the medical conditions our programs confront every day. The pandemic was just the most striking recent example of the good effects of getting this information-transfer right, and the terrible consequences of getting it wrong. We've seen this movie before, in preventing and treating cancer, diabetes, heart disease, maternal and infant mortality, and a host of other conditions for which we have good preventive and therapeutic options to teach about to save lives (and huge costs) by handling that information transfer well, or lose lives and treasure if we do it ham-handedly.  It's been estimated that by early 2022, the risk of illness and death from Covid was determined more by the flow of information than by the biology of the virus. That sounds dramatic, but it’s probably been equally true for years for the other conditions noted above. The pandemic was like a controlled experiment: when confronted by the same infectious agent (more or less) and the same preventions and treatments, some health care systems and communities did much better than others. True, the problem was often lack of access; but another major cause was lack of good transmission of accurate knowledge. This should be a bracing call to action for academic detailing programs as well as the healthcare delivery system as a whole. If the effective dissemination of accurate, actionable facts can play such an important role in infectious disease, cardiovascular illness, diabetes, pediatrics, obstetrics, and nearly every other branch of health care, the good news is that the “pop quiz” of Covid that we’ve just lived through can remind us how much we can do to enhance that flow of education to transform the best science into the best practice – as well as the awful consequences of getting it wrong. Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation! Biography.
Jerry Avorn, MD, Co-Founder & Special Adviser, NaRCAD Dr. Avorn is Professor of Medicine at Harvard Medical School and Chief Emeritus of the Division of Pharmacoepidemiology and Pharmacoeconomics (DoPE) at Brigham & Women's Hospital. A general internist, geriatrician, and drug epidemiologist, he pioneered the concept of academic detailing and is recognized internationally as a leading expert on this topic and on optimal medication use, particularly in the elderly. Read More. By Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD An interview with José Peña Bravo, PhD, Health Educator, Florida Department of Health in Duval County. Tags: Detailing Visits, Evidence-Based Medicine, Opioid Safety, Data  Anna: Hi, José! Thanks for joining us on our DETAILS blog today. Your journey that led you to the academic detailing community is unique – can you share that journey with us? José: Yes! My background is not originally in public health. I’m a biomedical researcher by training, specifically preclinical research using animal models. My dissertation work was on understanding the neurophysiological changes and different brain regions involved in behaviors related to substance use. My intention was to stay in academia and start my own lab, but my plans changed during COVID-19 and the opportunity to work with the public health department in Duval County presented itself. It’s been a learning curve for me to switch my perspective from preclinical research to public health—it’s been an enjoyable journey so far! Anna: I’m sure your biomedical research skills have a positive impact with clinicians during your detailing visits too, especially when clinicians want to discuss the neurobiology of substance use disorder. Speaking of visits, your detailing work is funded through CDC’s Overdose Data to Action (OD2A) grant, which seeks to prevent overdoses. Can you tell us about what the work for this grant looks like in Florida?  José: The OD2A project is a team effort across three CDC-funded jurisdictions in Florida. The health departments share that funding with various community organizations, and we all work toward linking patients with substance use disorder to treatment, mental health care, and care coordination services. Our detailing team is closely connected to organizations and resources within our community, and we share these resources with clinicians during our detailing visits. We also have access to aggregate prescription data from our jurisdiction and are continuing to find ways to present and incorporate this data at our visits with clinicians. We share this data and other resources across our three jurisdictions. Anna: We’ve found that many AD programs have been successful when they are closely connected to community resources. NaRCAD recently hosted a detailing training for OD2A recipients that you attended. What was it like to train with other jurisdictions working on the same project? José: It was helpful to hear from other jurisdictions because they’ve all approached their AD work differently based on the gaps in care in their own communities. I was able to hear from AD programs in rural areas and the specific challenges that their patients face with lack of access to care (long travel times, stigma, etc.). I also enjoyed practicing my detailing skills in a space where I felt comfortable making mistakes. It’s valuable to try things out and see how they’ll go before going out in the field. I learned a lot at the training and am excited to try out some of my new skills at my next visit.  Anna: Hearing from other detailers who are doing this important work with you is so helpful as you continue to think about and grow your own program. What advice would you tell other detailers working on the OD2A project? José: If you’re just starting out, reach out to community partners and get a sense of what patients with substance use disorder are experiencing and the challenges they’re facing before you start detailing clinicians. You’ll better be able to represent what is happening in the community and the resources that exist when you’ve done your research first! Anna: That’s terrific advice – a key piece of being an effective detailer is understanding the patient experience for the clinical topic you’re detailing on. So, what’s next for your work and Duval County? José: We’re currently working with our epidemiology team to collect population-level data and present it concisely. We want to be able to efficiently share this data with clinicians in a way that gets their attention and has them compare it to what they’re experiencing in their clinics to ensure an interactive dialogue during detailing visits. Anna: Using data to tell a story helps clinicians see the impact that they have in preventing overdoses and starts a conversation about organizations and resources that exist within communities for patients with substance use disorder. Thanks for sharing your OD2A work with us, José. We look forward to connecting with you and the other OD2A recipients at our conference in November! Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. José’s background is in Neuroscience preclinical research with over 10 years of experience in the field. His graduate work focused on the study of rodent models of substance abuse and the neurophysiological changes associated with controlled-substance experience. He has additional experience as an undergraduate and graduate level lecturer in different biomedical research topics. José recently transitioned to a position as health educator as part of the Overdose Data to Action (OD2A) program at the Florida Department of Health in Duval County. His role involves the implementation of the academic detailing program including outreach to clinics, integrating novel data and information to education materials, and keeping track of different metrics associated with outreach and AD sessions. By Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD An interview with Lexie Hach, Regional Health Specialist, Bureau of HIV, STD, and Hepatitis, Capacity Extension Program, Iowa Department of Public Health. Tags: Detailing Visits, Evidence-Based Medicine, Stigma, Rural AD Program  Anna: Hi, Lexie! We’re excited to chat with you today about your detailing work in rural Iowa. Your program started about 5 years ago and was born out of the HIV and Hepatitis Community Planning Group (CPG). Can you share more about the CPG and how the detailing program was created? Lexie: The CPG is made up of healthcare providers, people with lived experience, case managers, educators, and other stakeholders. The group serves in an advisory capacity to the Bureau of HIV, STD, and Hepatitis at the Iowa Department of Public Health (IDPH) and their main task is to ensure that the state has an inclusive participatory planning and evaluation process for the delivery of prevention and care services. About 5 years ago, the CPG recognized a need for more education in the rural parts of Iowa related to best practices in HIV, sexually transmitted infections (STIs), and Hepatitis. That’s when we learned about AD. We started building our detailing program based on a lot of the work that New York City’s program was doing. We now have 5 detailers, who we call Regional Health Specialists (RHS).  Anna: What an interesting start to your program! You’re currently assigned to detail in 15 counties in central Iowa covering over 20 sexual health topics, from stigma to your statewide condom program to new screening recommendations. Do you detail on these topics beyond the primary care setting? Lexie: Yes! Our goal as detailers is to provide the best health outcomes for people living with HIV and those living with, or at risk for, STIs and Hepatitis C. We meet with the medical community including, local health departments, community-based corrections, substance use specialties, mental health, and many community based organizations. We also attend a variety of community meetings. We know that many people living with HIV pass through community-based organizations. We want to make sure that those organizations are equipped with the correct information to get people the care they need through resources or referrals. Our team has created detailing materials for both medical professionals and community-based organizations to maximize our impact. Anna: Working with community-based organizations is a great way to make sure people have access to the best possible prevention and care, especially in a rural state like Iowa. How does Iowa’s geography impact your detailing work?  Lexie: Iowa has 3.1 million people with about 54 people per square mile. Our state is 85% farmland with country roads, winter weather, and a lot of construction. It is not uncommon to lose cell service or your GPS signal while driving in the very rural parts of Iowa. We’ve adapted our detailing approach because of this and have found that group sessions work best for us. We identify champions in the clinics and are then able to set up 1:1 visits with clinicians as needed. Anna: We often hear from programs that group detailing is a great way to get your foot in the door and spread the word about AD. It’s nice to know that it’s working so well for your program too! I imagine Iowa’s geography also impacts patient care. Lexie: Yes, it does. There can be stigma around seeking sexual health care in rural parts of Iowa. Many times, patients know someone working at the clinic in their town. It’s common for patients to travel long distances to get to a clinic where they don’t know anyone. It’s also common for patients to have to travel over 2 hours to access HIV care at a specialty clinic. Anna: And that’s 2 hours one way! Has your program been able help with increasing access to care? Lexie: Yes, I can share one story that stands out the most to me. A couple of years ago, I was presenting at a community meeting with 15 or so people in attendance. The CEO of a federally qualified health center (FQHC) in Southern Iowa was also present and we were able to connect and network afterwards. We talked about how far patients must travel for Ryan White HIV care and he saw a need that the community health center could fill. We connected with senior leadership, there were additional meetings, and together we were able to bring Ryan White HIV care services to the health center. Those services are still there today and now people living with HIV in Southern Iowa can access Ryan White services closer to home. Anna: There is so much to be said about the power of AD, networking, and champions! Let’s wrap up with hearing about some of your program’s successes from this past year. Lexie: We’re so proud of all that we’ve accomplished this year! Here are some highlights: Anna: Those are some impressive numbers across both the clinical and community settings. Thank you for letting us feature your detailing work and learn more about the impact your team is making in your state! Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. Lexie Hach holds an Associate Degree in Nursing, Bachelor of Arts degree in Health Promotion and Women’s Health, and a Master of Arts degree in Community Health Education. Lexie is currently a Doctoral student in Public Health (DrPH) with an Epidemiology concentration at the University of Nebraska Medical Center (UNMC). Lexie has held positions in nursing, health education, public health, and healthcare marketing. Lexie has been with the Bureau of HIV, STD, and Hepatitis Capacity Extension Program at the Iowa Department of Public Health for over five years as a Regional Health Specialist (RHS). Lexie was previously with the bureau as a Disease Intervention Specialist (DIS). Lexie was instrumental in developing and implementing the RHS program in Iowa’s rural communities. In her RHS (academic detailing) role, Lexie educated a variety of different stakeholders on best practices related to HIV, STIs, and Hepatitis in central Iowa. Lexie has helped support the Sexually Transmitted Disease (STD) Program as a DIS due to COVID-19 and also helped as a COVID-19 contact tracer. Lexie was the previous workforce health department co-chair for Iowa’s statewide strategic plan to stop HIV in Iowa (Stop HIV Iowa). Lexie recently returned to her roots in the STD program and now serves as an Integrated DIS and Special Projects Coordinator, but continues to work alongside her fellow RHS colleagues. By Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD An interview with Sandeep (Sonny) Singh Bains, PharmD, BCPS, Clinical Educator, Alosa Health. Alosa Health is a national leader in developing and implementing academic detailing programs to improve prescribing. Tags: Detailing Visits, Evidence-Based Medicine, Primary Care, Training  Anna: Hi, Sonny! It’s wonderful to connect with you today and hear about your work as a clinical educator at Alosa Health. What has your clinical pharmacist journey looked like to date? Sonny: I’ve been working for over a decade in hospitals and primary care. I completed my residency in hospital pharmacy, and as I continued to do administrative and clinical work in hospitals, my interests shifted more towards primary care and population health. I’m originally from California where it’s common to have pharmacy involved in primary care. When I moved to Pennsylvania, a lot of clinicians didn’t understand why a pharmacist would be in a primary care setting and were skeptical of me and my role within the clinic. Fast-forward 10 years, and almost every health system in Philadelphia has a pharmacist on their team within their primary care network. I’d like to think that I can take a little bit of credit for paving the way for pharmacists on primary care teams throughout our state! Anna: That’s definitely something to be proud of. How did you end up being a detailer?  Sonny: I’d been noticing a lot of overlap in the work being done in our primary care clinics by different members of the team. I ended up creating protocols to automate certain things for different chronic conditions and people would say, “Wow, this is brilliant.” It really wasn’t brilliant; I was using the same set of guidelines in the literature I was reading and was communicating that to clinicians to make workflows easier. I didn’t know about the strategy of academic detailing at the time, but I knew I needed to educate clinicians. I was fortunate enough to be connected with Alosa Health and I now work for them as a full-time consultant. I work as a detailer and also help hire, interview, train, coach, and mentor new detailers on the team. Anna: You were meant to be a detailer before you even knew what it was! Alosa has been working on diverse clinical topics over the years including, antiplatelets, atrial fibrillation, immunizations for elderly, serious illness conversations, chronic obstructive pulmonary disease (COPD), heart failure, dementia, atherosclerotic cardiovascular disease (ASCVD) prevention, diabetes, acute pain, chronic pain, and opioid use disorder. What have been some of the campaigns where you’ve felt you’ve made an impact? Sonny: It’s a lot of fun to be working on so many different campaigns. Many of the topics fit right into my wheelhouse with my hospital and primary care background. Diabetes has been my favorite topic. I’ve been able to make a large impact for patients and care providers. We’re able to improve lives of patients (prevention of stroke/heart attacks) and improve outcomes for providers and health systems (financial incentives, quality ratings, etc.). I’m also proud to be part of the pain modules. The opioid epidemic has impacted all of us in America and I’m glad to be able to do my part in helping to bring resources and evidence-based treatment to local providers in our community.  Anna: It sure sounds like you’ve been able to make an impact! How has your work with Alosa evolved over the years? Sonny: Alosa Health has been expanding and we have several new partnerships and collaborations throughout the United States. We started detailing only in the state of Pennsylvania and we’ve now expanded to numerous states. It’s been exciting to be part of the leadership team to help coach detailers, as well as create creative partnerships with local health systems to improve outcomes for patients. Anna: What challenges have come up as your program has continued to expand? Sonny: The biggest barrier we’ve experienced, like many detailing programs, has been access to clinicians and the ability to set up visits. I’ve had relationships with clinicians and clinic staff in the past where I could have walked into any office at any given time, but I’ve lost a lot of those relationships with COVID and staff turnover. I’ve been working hard to reestablish relationships in the field and teach new detailers how important strong relationships are to our work as detailers.  Anna: What approaches have you taken to reestablish relationships, as well as to gain access to new clinics? Sonny: I like to partner with colleagues that I’ve worked with previously. I recently connected with a former colleague who’s a VP at a large healthcare organization and we quickly realized we could help each other improve patient outcomes and reduce cost. From this connection, I was able to get buy-in at the administrative level and accessed hundreds of prescribers. They even gave their clinicians a monetary incentive to meet with our detailers. We’ve also benefited from connecting with administrators of health systems. We approach them like business partners; it takes a special skill to communicate and work with an administrator. It’s important to have team members who understand how to communicate with leadership effectively to get that buy-in. Anna: It might take more time to get buy-in from administrators, but it opens so many doors once it’s done successfully. As we wrap up, can you tell us some tips to help our readers be effective detailers and communicators? Sonny:
Anna: It’s so important to put the work in to build solid relationships with clinicians in order to be an effective detailer. Thanks for joining us on the DETAILS blog, Sonny - our AD community will gain a lot from the insights you’ve shared today! Learn more about Alosa’s work: alosahealth.org/clinical-modules Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. Sandeep is a clinical pharmacist with extensive clinical leadership experience in both acute care and ambulatory healthcare settings. He completed post-graduate training at Einstein Medical Center in Philadelphia and has since worked in clinical pharmacy leadership with large healthcare systems in the greater Philadelphia area. He is also the principal consultant at Bains Rx, LLC, a healthcare consulting firm dedicated to improving outcomes while reducing costs. By Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD An interview with Adrienne Butterwick, MPH, CHES, Senior Improvement Advisor and Academic Detailing Project Manager, Comagine Health. Comagine Health is a national, nonprofit, health care consulting firm that works collaboratively with patients, providers, payers and other stakeholders to reimagine, redesign and implement sustainable improvements in the health care system. Tags: Detailing Visits, Evidence Based, Substance Use, Opioid Safety  Anna: Hi Adrienne! We recently saw you present on a panel where you spoke about your academic detailing project with dentists on opioid safety. Can you tell us a little more about how your team got started with this work? Adrienne: In 2018, the CDC released funds to states through the Overdose Data to Action (OD2A) grant and the state of Utah selected academic detailing as one of the interventions they wanted to use. AD is one of the many different modalities that we use within my organization to reach clinicians to educate them and have an impact on the kind of care they provide. The state began looking at specific regions and populations to target after we received the funding. Utah is unique in that it has a high number of adolescents undergoing surgery for wisdom teeth removal, which is one of the most common instances where controlled substances are prescribed. A first prescription can be a huge turning point to potentially becoming addicted to a substance, especially at a young age. That’s when we decided to put together a team of two detailers to detail dentists. I was lucky enough to attend each detailing visit and collect data through pre- and post-surveys and answer any administrative questions that came up.  Anna: It’s impressive that your organization was able to look at the data in your state and build a program to fill a specific care need. What makes dentists and their environments unique when it comes to detailing? Adrienne: There’s a theory that providers who are prescribing controlled substances are working within systems and teams that are well-poised to understand the challenges of opioid prescribing. Dentists fall into a different healthcare model that’s often siloed; they aren’t usually affiliated with an overarching health system or university like many primary care providers are. This results in isolation, making the interactive, 1:1 outreach model of detailing even more important – we knew we needed to bring the information and support directly to them in their dental offices. Anna: Detailing seems like a critical need for isolated dentists, both in providing them with customized education, but also in building connections. Were there any special considerations that your team took into account as you worked with the dentists? Adrienne: The language that’s used in the dental world is very different than language that’s used in primary care. We were fortunate enough to have a dental provider, who’s a champion of AD, work with us as a detailer on our project. He knew the language, understood the workflow, and could speak to the need for safe opioid prescribing. He always started his detailing sessions with a personal story like, “When I took wisdom teeth out, I would always prescribe 40 Percocet pills. All I can think of today is, ‘what have I done?’” You could see the mood shift the moment he started talking about his personal experiences, allowing for a connection between himself and the dentists he met. The success of this program wouldn’t have gone even half as far without his support.  Anna: A detailer who can build empathy with clinicians and who has personal experience with a challenging topic is an important asset to have in a detailing program. What obstacles did you face as your team implemented this project? Adrienne: Connecting with dental offices, in general, was tough. We first started by working with dental associations to get relationships in place. We submitted newsletter articles, attended meetings, presented at the regional conference, and sent our program’s information via their listservs. We also Googled practices and found ones that had more than one dentist working in the office at a time. We’d cold call those offices and say, “It looks like you have a big operation – is there a way we could bring training in for your team for continuing education credits?” Before leaving the visits, we’d ask the dentists for referrals to other clinicians and leave flyers behind. Relationships grew organically over time. Anna: It sounds like the project began to build on itself fairly quickly. Did your team experience any barriers from the dentists during the detailing visits? Adrienne: We had a lot of dentists who thought the opioid crisis wasn’t relevant to their practice and we knew that we had to find ways to tie it into their profession. Fortunately, dentists have historically been involved in public health movements because they hold a different type of relationship with patients that is closer than a typical relationship with a primary care provider. They see patients more frequently and can detect small changes in health quickly. The dental profession was incredibly important in the tobacco cessation movement in the 1990s. They were instrumental in getting individuals to reduce or completely stop using tobacco. Dentists are also starting to be trained in domestic violence and human trafficking. For the dentists who were hesitant about the relevance of our detailing visits, we would say, “You have this amazing relationship with patients that we don’t see in other parts of healthcare—here’s how you can make a huge difference!” or “I can understand how there would be a lot of fear to step out of your comfort zone; we have a lot of resources and materials to support you.”  Anna: Dentists truly have a unique relationship with patients that can be used to promote countless public health initiatives. Can you think of a time your team was able to empower a dentist to change behavior and encourage them to see their relevance in combatting the opioid crisis? Adrienne: There was a dental group in a rural part of the state that had one dentist and a big support staff. We came in for a detailing visit and had a conversation with the entire office. After the meeting, one of the dental assistants pulled me aside and told me that a patient who had recently completed substance use rehab had visited the office in need of a procedure that would warrant prescribing an opioid. No one in the office knew what to do for pain control and they were all unsure how to approach the patient given his history. She said that because we came, she felt like she now knew how to have a conversation with him about the procedure and his safer, alternative options for pain management. The dentist also shared that prior to our visit, he often didn’t know how to handle conversations about pain management and opioids and wasn’t sure if it was his job to do so. After our visit, he said he felt comfortable and confident doing this, and shared an anecdote of being able to create a safe space for an ongoing conversation with a recent patient. Anna: It seems like your team has had such an impact by using one of the core elements of detailing – building relationships through empathy, validation, and support. Can you share some encouragement for readers who are considering having these conversations with dentists? Adrienne: Be flexible and don’t come in with your own agenda – be sure to let the dentists drive the conversation and let them teach you along the way. It can be a rewarding yet challenging experience – don’t forget to celebrate the small wins on your journey! Anna: Thanks for sharing this innovative approach to detailing, Adrienne! We’re looking forward to hearing about your continued impact with the dental community and beyond. Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. Ms. Butterwick is a Senior Improvement Advisor at Comagine Health. She is currently working on quality improvement efforts directed by the Centers for Medicare & Medicaid Services (CMS) to improve quality of care for residents living in post-acute and long term care as well as assisted living and home health. She's also working on an initiative to increase advance care practices in those settings. In addition, through a subcontract with the Utah Department of Health, Ms. Butterwick currently provides educational support for opioid prescribing to family medicine and dental providers. Her work with this contract has earned national recognition and has been presented at the RX Drug and Heroin Abuse Summit in April 2020 and the American Public Health Association’s annual conference in October 2020. She is currently also collaborating with faculty from the University of Utah regarding telehealth and advance care planning initiatives through the Utah Geriatric Education Consortium and Geriatric Workforce Enhancement Programs. She completed her Bachelors of Science degree in Behavioral Science and Health at the University of Utah in 2007 and her Master's in Public Health at Westminster College in 2014. She has also earned recognition as a Certified Healthcare Education Specialist (CHES). In her 15 years of public health project management she has also worked in rural health research, provider education programs and care management. She has a strong passion for quality improvement and public health.  Words of AD Wisdom is a series that features tried-and-true practices from AD experts. Our guest blogger this week is Chirag Rathod, PharmD, a NaRCAD Facilitator and Academic Detailer at Illinois ADVANCE. Tags: Detailing Visits, Evidence Based Medicine, Materials  Simplify your messaging! Prior to your detailing visit, think about ways to slow down and limit the amount of information you share with the clinician so that the visit is of most value to them. You can do this by asking focused needs assessment questions and providing the clinician ample opportunities to speak and engage in the conversation.  Focus. Focus. Focus. Resist the urge to cover too much content in your detailing aid during an AD visit. A more focused discussion on one section of your brochure can be more fruitful than trying to cover every key message during a single visit.  Open-ended questions will set you free! There is limited usefulness in asking closed-ended questions (e.g., confirming something about the clinician’s practice). The use of open-ended questions can help you gain layered insight into the clinician’s practice and tailor the visit to them. Asking open-ended questions allows opportunities for the clinician to speak about their experiences in detail and for you to listen and deliver supportive, relevant key messages.  Prime your “AD Kitchen.” In cooking shows, hosts show you how to prep a meal, pulling out the final, cooked meal from the oven in the following scene. For an AD visit, you should think about how you can prep your ‘ingredients’ for clinicians by making sure you’re well-versed in your materials and are ready to teach your clinicians how to adopt something practical and usable into their practice. Have a tool to share? Test it in advance of your visit, so you can confidently demonstrate effective use of the tool, leaving more time to discuss applying the tool successfully in practice with patients. Want more tips? Stay tuned for the next installment in our Words of AD Wisdom series, and reach out to the NaRCAD team, subscribe to our network, or check out our discussion forum to hear more tips and ways to prime your AD kitchen for an efficient visit!  Biography. Chirag is a clinical pharmacist, academic detailer and instructor with Illinois ADVANCE at the University of Illinois-Chicago (UIC). He graduated with a Doctor of Pharmacy degree from Midwestern University in 2012 and has been working at UIC ever since. During his time at UIC, he has been involved in a number of collaborations with the College of Pharmacy, College of Dentistry, and College of Medicine in addition to teaching Medical Improv, which utilizes the techniques of improvisation to improve communication skills amongst current and future healthcare practitioners. He has also been focused on academic detailing, including program planning, presentations to recruit organizations, creating educational material, training staff, facilitating role plays and providing individualized feedback, and detailing prescribers. He trained with NaRCAD in 2019 during their Spring Training Series to develop his skills as an academic detailer and has also presented at the NaRCAD annual conference. His interests include performing improv, podcasting, sports and hanging out with his sister’s dog. Fun fact: Chirag hiked Mount Kilimanjaro in February of 2022. Supporting Clinicians in Utah: Working Together to Utilize Safe Opioid Prescribing Guidelines3/25/2022
An interview with Parveen Ghani, MBBS, MPH, MS, Health Program Specialist III, Division of Professional Licensing, State of Utah. by Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD Tags: Opioid Safety, Evidence Based, Training  Anna: Hi Parveen! You’re one of our training alumni who’s built a strong program over the past few years. We’re thrilled to be able to catch up with you! Can you tell us about yourself? Parveen: I’m trained as a physician and have always wanted to work in public health. It was important to me to be able to make a difference in people’s lives. I currently work in the Division of Professional Licensing at the Department of Commerce in Utah. I've been working as an academic detailer since my NaRCAD training a few years ago. Anna: It sounds like the rest is history! Are there other detailers on your team who are helping you meet your program goals? Parveen: I’m a full-time detailer for our AD program along with my colleague, Marie Frankos. We work with many of the same prescribers over multiple detailing visits and build strong connections with them.  Anna: Can you talk to us about your detailing work in overdose prevention? Parveen: Opioid overdose in the State of Utah is exceptionally high. We’re currently working with prescribers on the safe prescribing of opioids. Our state’s prescription drug monitoring program is called the Controlled Substance Database Program (CSD). The CSD includes both a Patient Dashboard and Prescriber Dashboard. The Patient Dashboard is an electronic clinical decision-making tool that grants prescribers access to information regarding controlled substance prescriptions for individual patients. It contains records of a patient’s poisoning or overdose and any violations associated with a controlled substance. The Prescriber Dashboard, on the other hand, tracks each clinician's prescribing patterns and CSD utilization behavior. Anna: We’ve seen a lot of success with detailing programs who work with clinicians to navigate their state’s prescription drug monitoring program, like your CSD. Does your state require prescribers to look at this database?  Parveen: Yes. According to the Utah Controlled Substances Act, (a) A prescriber shall check the database for information about a patient before the first time the prescriber gives a prescription to a patient for a Schedule II opioid or a Schedule III opioid. (b) If a prescriber is repeatedly prescribing a Schedule II opioid or Schedule III opioid to a patient, the prescriber shall periodically review information about the patient in: (i) the database; or (ii) other similar records of controlled substances the patient has filled. Anna: It’s so important to support prescribers in using a database like this, especially when there are mandates in place. What is the overall goal of your AD program? Parveen: The goal of our AD program is to provide recommendations to prescribers regarding best practices in the utilization of the CSD per the Controlled Substance Database Act. This includes identifying individual prescriber’s prescribing and dispensing patterns of controlled substances, identifying prescribers who are prescribing in an unprofessional or unlawful manner, and identifying polypharmacy, doctor shopping, poisoning, or overdoses. Anna: It sounds like your AD program is working hard to support clinicians in CSD utilization. What kind of resources have you developed for clinicians that work towards your program’s overall goal, and how do you make these materials accessible?  Parveen: We’ve created a toolkit that acts as a guide to help clinicians utilize the database and different resources within the community. During our in-person visits, we provide hard copies of materials that include screenshots of how to create a CSD account, reset CSD account passwords, and navigate the dashboards within the CSD. During our virtual AD sessions, we send these materials electronically. Additionally, we provide our contact information for further technical assistance, including our personal phone number, work phone number, and email address. We've made our toolkit available on our website along with prescriber FAQs. We’re continuing to update our website with helpful materials for clinicians. Anna: Making resources like this so accessible is key. Can you share some reflections on visits where you felt like you made a difference or were able to offer technical assistance? Parveen: I love helping prescribers, even if it is something as simple as walking them through the log-in process or resetting a password. I’ve had clinicians bring their entire medical team in for a detailing visit so that I can show everyone in the office how to use the database. One prescriber even told me after a visit that they would be sharing my name with a colleague and that I should expect a call to schedule a detailing visit. It’s lovely to get these types of referrals from the clinicians. Anna: Prescribers feeling thankful and impressed with your 1:1 support enough to refer you to their colleagues is a huge success! Let’s wrap up with one more question - what’s one tip you’d give to another academic detailer? Parveen: Find ways to collaborate. We can’t do it alone! Start working together with other programs and share information, especially community resources. We can really make a difference if we work together. Anna: I couldn’t agree more. Making community connections and sharing information allows for great success in accomplishing goals for both small and large initiatives. Our AD community will be able to glean a lot from your program’s successes, and we look forward to sharing more of your team’s expertise in the future. Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. Parveen Ghani has over eight years of work experience in public health. She obtained her Master in Public Health degree (MPH) from Walden University (Minneapolis, Minnesota). Following this, she worked for four years with the Office of Minority Health for the Nebraska Department of Health and Human Service. Parveen relocated to Idaho Falls in 2015 with her husband and began to pursue her career in bioinformatics. She obtained her master’s degree in Biomedical Informatics from the University of Utah in May 2018. Shortly after graduation, she started working as an Academic Detailing Specialist with the Division of Professional Licensing (DOPL), Salt Lake City, Utah. Before moving to the United States, Parveen earned her medical degree (MBBS) from Dhaka Medical College, Bangladesh. While not licensed in the United States, Parveen has worked as a physician in Bangladesh, Ireland, and Australia. Parveen enjoys working with the prescribers on the safe prescribing of opioids. Parveen loves to exercise, walk, read, play the piano, and play with her pet kitty in her leisure time. An interview with Carolyn Wilson, a Senior Health Program Coordinator at Ledge Light Health District. Ledge Light Health District is located in New London, Connecticut and is the regional health district serving the southern part of New London County. by Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD Tags: Opioid Safety, Evidence Based Medicine, Substance Use  Ledge Light Healthwatch Ledge Light Healthwatch Anna: Carolyn, we’re thrilled to feature you on our DETAILS blog! I know you wear many hats – can you tell us about your current job role? Carolyn: I’m a health educator working within primary prevention, an academic detailer, and the host of our health district’s television program called Healthwatch. Healthwatch covers topics like mental health, physical health, disaster preparedness, general public health, COVID-19, environmental health, and disease prevention. I’ve been with Ledge Light Health District for 11 years. Anna: It seems like improving patient and community health outcomes is a common thread across all your roles. What primary prevention work or related projects complement your AD work? Carolyn: Depending on what topic I'm detailing on, I lean into my primary prevention work or the harm reduction work that my colleagues are working on. One of the larger initiatives I often share with clinicians during detailing visits is the Naloxone and Overdose Response App (NORA) project. The Department of Public Health developed a web-based application that can be downloaded directly to your phone. It has information about preventing, treating, and reporting opioid overdose. The app can be used by both folks in the community and clinicians. I also speak to clinicians about proper medication storage and disposal while promoting our “Take it To The Box” Initiative.  Anna: We love to see programs using AD to spread the word about broader, community-focused initiatives. Are there other ways that your opioid-related AD work overlaps with work being done within your department? Carolyn: Yes! I’m so lucky to be able to work in the office side-by-side a recovery navigator. She helps link folks in the community to addiction services. Every day we say things like, “hey, I overheard you talking to that pharmacist just now – do they know x clinician?” We often share resources and try to work together to ensure that community health goals are achieved, often by making sure that the work people are doing is connected rather than existing within silos. It all comes down to helping one another work towards a common goal. Anna: What better way to work towards a common goal than to share resources across colleagues and projects! Can you share a story from the field where there was an intersection among various projects?  Carolyn: I detail a lot of advanced practice nurses (APRNs) and also work with them on some of my primary prevention projects. The overlap in projects helps me build strong relationships with these clinicians. I sometimes work with school-based health centers as part of my prevention work, and these health centers are typically run by APRNs. These centers act as an access point to care for many students and families. It’s essentially a primary care clinic right in the school. The Child and Family Agency oversees the school-based health centers in southeastern Connecticut and reached out to me after a horrific event in a Connecticut middle school. A few months ago, a 12-year-old got access to fentanyl and brought it to school. He overdosed and passed away a few days later at the hospital. We haven’t seen many overdoses in schools, but after this happened, a lot of schools started looking at their policies and school-based health centers wanted to have naloxone on hand. The medical director of the Child and Family Agency advocated for a policy that required all school-based health centers to have naloxone and to be trained in administering it. Anna: What a devastating story. Have the school-based health centers been able to put these types of new policies into place? Carolyn: When one of the clinicians from the Child and Family Agency reached out to me, she said, “Carolyn, I know you do this kind of work. You trained me in naloxone not too long ago during an academic detailing visit. I’d like to have a naloxone training for my nurse practitioners in the school-based health centers. I want naloxone available in all of our clinics.” This type of request would typically be delegated to somebody else in our department, but because of the relationships I had built through academic detailing, I was asked to provide the training, and I did. As a result, the school-based health centers now all have access to naloxone and the clinicians know how to administer it.  Anna: It’s incredible that you’d built trusting relationships with clinicians enough to be asked to provide this training, contributing to changing a policy in a span of one or two months. Carolyn: It means a lot that they came to me because they trusted me and knew I could get it done for them. I truly don't think I would have been involved if it wasn’t for my academic detailing work. Anna: I agree. It’s been a pleasure learning about your work and your unique approach to academic detailing. We’re excited to follow along with you on your AD journey as you continue to promote health across your community. Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. Carolyn Wilson is a health educator and prevention specialist serving as a program coordinator at Ledge Light Health District in New London CT for 11 years. Carolyn studied public health and health education at New York Medical College. Keenly interested in health promotion and behavioral science, Carolyn enjoys bringing her passions and talents to both primary prevention and academic detailing work. Carolyn has been serving as an academic detailer for over 2 years and enjoys speaking with clinicians about strategies to prevent opioid related deaths. Carolyn also manages the Groton Alliance for Substance abuse Prevention @Groton_Prevents. In her spare time, Carolyn enjoys serving on the Board of Directors for the CT Association of Prevention Professionals and Fiddleheads Food Cooperative. To connect with Carolyn, find her on LinkedIn. The Impact of Childhood Experiences on Patient Health: AD to Encourage Trauma-Informed Care1/31/2022
By Anna Morgan-Barsamian, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD  As we’ve jumped right into 2022 programming, our team at NaRCAD continues to support detailing efforts that improve outcomes for vulnerable populations, recognizing that patient needs are complex and often informed by social determinants of health. One critical topic for which we’ve developed resources focuses on detailing to improve patient health as informed by both adverse and positive childhood experiences. We’re seeing an increased need from our community members to support detailing efforts related to this critical topic. We partnered with the National Association of County and City Health Officials (NACCHO), Centers for Disease Control and Prevention (CDC), and consultants from Brigham and Women’s Hospital and Tufts Medical Center over the past two years to develop academic detailing materials for clinicians working with both adults and children. The CDC defines adverse childhood experiences (ACEs) as traumatic events that occur in childhood, including abuse, neglect, and family dysfunction. These events are linked to poor health outcomes in adulthood like chronic health problems, mental health conditions, and substance use. When considering the impact of environment on health, the CDC notes that children are likely to flourish when they have safe, nurturing relationships. These supportive settings create the opportunity for positive childhood experiences (PCEs). PCEs can mitigate the effects of ACEs and toxic stress, promote healing from traumatic events, and foster healthy development and learning in children.  Encouraging clinicians to consider both ACEs and PCEs is a natural fit for clinical outreach educators. There are many reasons that clinicians may not be prepared to have conversations with patients about childhood experiences. Perhaps the clinician doesn’t have a behavioral health provider to consult within their practice, so they don’t know who to turn to if a patient discloses a traumatic experience. A detailer can encourage a clinician to explore this concern, as well as provide a list of local behavioral health providers in the community. Another barrier may be that clinicians could feel hesitant to approach discussions related to trauma, or may not have the skills to do so with sensitivity. In this case, detailers can offer tools that illustrate supportive language that creates a safe environment for patients to share their experiences. Supporting clinicians in using evidence-based tools to build trust with their patients signals that it’s not only okay to talk to patients about this sensitive topic; it’s vital for clinicians to lead these conversations in order to ascertain their patients’ needs and promote patient-centered decision making. A detailer can encourage clinicians to adopt new behaviors through specific key messages, including asking clinicians to take the following steps to support adult patients:
 When clinicians are supporting pediatric patients, detailers can deliver this set of evidence-based key messages, encouraging clinicians to:
 With evidence-based tools, successful AD interventions related to ACEs can result in multi-faceted support for patients, such as stronger connections with community resources, mental health supports, and more trusting relationships between clinicians and their patients. We hope to continue to explore this important and complex topic together as a community. If your program is currently working on an ACEs or PCEs AD campaign, or is interested in starting one, please reach out to us so we can support the development of your programming. We always love collaborating and learning more about the important work that you’re doing, and we hope to continue to build our resources and create toolkits in support of complex topics such as these that intersect with other behavioral health and prevention-focused AD campaigns. Have thoughts on our DETAILS Blog posts?
You can head on over to our Discussion Forum to continue the conversation! Academic Detailing to Address Substance Use in New Jersey: An “All-Hands-on-Deck” Approach11/30/2021
An interview with Clement Chen, PharmD, BCPS, Clinical Pharmacist Specialist, Rutgers New Jersey Medical School. by Anna Morgan, MPH, RN, PMP, Senior Manager, Training & Education, NaRCAD Tags: Substance Use, Detailing Visits, Evidence Based Medicine  Anna: Hi Clement! We can’t wait to hear about all the impactful work you’ve been doing in New Jersey. Tell us a little about your academic detailing program. Clement: The Northern New Jersey Medication-Assisted Treatment Center of Excellence was established in 2019 and is funded by the New Jersey Department of Human Services. Our goal is to not only increase access to medications for opioid use disorder (MOUD) in office-based addiction treatment practices, but also to ensure that providers treat this chronic disease with evidence-based practices. Our vision is to end the stigma of addiction and ensure that all people with substance use disorders have access to high quality care so they may live full and satisfied lives. I was hired as the academic detailer to provide education and identify the needs of providers in these practices. Anna: Tell me more about the types of providers you visit. Clement: I’ve been working with providers from all areas of care. In addition to office-based providers, they also include those from hospital settings, psychiatric hospitals, community health centers, substance use licensed facilities, and prison settings. Addressing the substance use crisis requires an “all-hands-on-deck” approach. Any setting where patients seek care are environments where detailing can improve patient access to support. Anna: It’s so important to approach academic detailing with a wider lens and consider where it can fit in with other community initiatives. Can you walk us through some of the work you’ve done so far, or are planning to do, in each of these settings? Clement: I’d be happy to!  Hospital Setting: One of the inner-city hospitals that we are working with is looking to establish an addiction medicine service within their hospital system. The goal is to provide trainings and consultations on best practices for addiction medicine, in addition to technical assistance in setting up a program. I’ll be working with their attending physicians and residents to provide evidence-based practice trainings. Another one of our partners, an addiction medicine provider, has been pushing for greater initiation of MOUD in her own hospital system. The main issue is to ensure that providers use MOUD in an evidence-based manner and adhere to the latest findings. For example, the provider has been receiving pushback from other providers due to the lack of referral sources. These providers have shared that they believe MOUD should not be initiated without a “warm handoff.” Furthermore, buprenorphine has been discontinued during the pre and peri-operative settings and only resumed post-operatively despite growing evidence for continuing buprenorphine in these settings. I plan to detail these providers in-person to provide literature supporting the use of MOUD, even when warm handoffs are unavailable. I’ve provided supporting literature and a summary to the addiction medicine provider to assist with her case to expand this initiative, which helped her to develop an information sheet to justify the expansion of MOUD in her hospital. Psychiatric Hospital Setting: We’ve also partnered with physician champions at some of the psychiatric hospitals in New Jersey. Stigma and the inaccurate idea that patients do not experience acute withdrawal for opioid use has made many providers hesitant to start therapy with buprenorphine. Psychiatric hospitals have been fervently looking to provide treatment for opioid use disorders. The goal is to implement a clinic designed for initiating and maintaining those on buprenorphine and naltrexone extended release. We’ll be assisting with the implementation of the clinic and provide individual in-person detailing visits for the providers at the hospital. To prepare for this, we’ve developed several informational sheets on MOUD and other related information to give to the providers.  Community Health Center Setting: One of the community health centers that we’ve partnered with wants to begin providing MOUD for those already on the therapy. There are several advanced nurse practitioners and physicians at the health centers looking for more guidance and support on the appropriate prescribing of buprenorphine. I’ll be working with their team to provide them with evidence-based practices and help them with buprenorphine induction strategies. Substance Use Licensed Facility Setting: One of the substance use licensed facilities I’ve consulted with mentioned that fentanyl has made initiation of buprenorphine very difficult due to the increase in the number of cases with precipitated withdrawal. As a detailer, I’ve worked closely with their lead providers, providing them with available literature on alternative buprenorphine induction strategies. They're in the process of updating their protocols for the induction of buprenorphine. Another facility is working with us to start prescribing MOUD within their residential settings. Prison Setting: I regularly consult with the Department of Corrections providers since access to MOUD have traditionally been low in the prison system. Their patients also have unique needs compared to those in the community setting. I meet with them on a monthly basis via a case conference and discuss clinical solutions in order to help them provide the best care to their patients.  Anna: Your work is incredibly comprehensive and thoughtful. It’s truly amazing to see how your team has incorporated academic detailing into so many initiatives and clinical settings within your community. Clement: We believe that to overcome this crisis, all patients with opioid use disorder need equitable and timely access to care. With the record number of overdose deaths reaching over 100,000 in one year, this is our greatest focus. We’re confident that our academic detailing work will help us achieve this goal. Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Biography. Dr. Clement Chen graduated from the Ernest Mario School of Pharmacy at Rutgers University in 2013 with his Pharm.D. He then completed a one-year residency at the Hudson Valley Veterans Affairs Ambulatory Care Clinic. Thereafter, he has worked as a staff pharmacist at St. Michael’s Medical Center in Newark and as a cardiology heart failure clinical pharmacist specialist at University Hospital in Newark. In addition to working as a clinical pharmacist specialist, he was a Transitions of Care pharmacist at Hunterdon Medical Center from 2016-2020. This has helped him to balance his inpatient and outpatient roles as a pharmacist. To further demonstrate proficiency in clinical pharmacy care, he received his Board Certification in Pharmacotherapy in July of 2016. His current role is as the academic detailer in the Northern NJ Center of Excellence, with the goal of providing education and support to increase statewide capacity to provide medications for addictions treatment (MAT) for patients with substance use disorders with a focus on opioid use disorder. This interview features Carla Foster, MPH, who leads the conceptualization, implementation, and evaluation of Public Health Detailing as an Epidemiologist within the Bureau of Alcohol and Drug Use Prevention, Care and Treatment (BADUPCT) at the New York City Department of Health and Mental Hygiene (NYC DOHMH). She is currently activated for the COVID-19 emergency response as Lead Analyst managing the Reporting Unit within the Integrated Data Team of DOHMH’s Incident Command System. By Winnie Ho, Program Coordinator Tags: Data, Detailing Visits, Evidence-Based Medicine, Health Disparities, Program Management, Stigma, Substance Use, Training  Winnie: Hi Carla! You’ve certainly had a lot on your plate with so many diverse campaigns. Can you walk us through the conceptualization process for your detailing campaigns, and how your team came to choose cocaine use as your current detailing topic? Carla: We can start with some data on this. In 2018, more New Yorkers died from drug overdose than from homicide, suicide, and motor vehicle crashes combined. Cocaine – in both crack and powder forms – has played an increasingly prominent role in this crisis. The mortality rate from overdose deaths involving cocaine more than doubled between 2014 to 2018, amounting to 52% of all drug overdose deaths in NYC. Some of the associated risks are serious - increased exposure risk to fentanyl, cardiovascular disease events and death.  W: That’s stunning data. Especially in the midst of the opioid crisis, it’s important that we don’t lose sight of other substance use issues going on right now. I’d love to learn a little more about the challenges and lessons that your team has learned by detailing on cocaine use. C: First, we have to be aware that fentanyl, a powerful opioid 50 to 100 times stronger than morphine may be found in many substances, including cocaine. We’re very concerned about fentanyl and cocaine because people who use cocaine do not have tolerance to opioids and are at even higher risk for overdose. It’s also important to address the perception of who is most impacted by high mortality rates. There’s this idea that cocaine use is more prominent in younger populations, but our data show that it’s actually impacting an older population more than many might expect. In particular, residents age 55-84 in the Bronx Borough have experienced the largest increase in cocaine overdose death rates in New York City from 2014 to 2018.  That’s why it’s critical for us to raise awareness in an effort to mitigate misconceptions and stigma around risky use and those who may have a substance use disorder (SUD). In addition to shame, there are still very real potential socioeconomic and legal consequences from disclosing substance use, which can deter folks from even seeking help. We take into account the unjust consequences of policies applied unevenly according to race, and how this impacts implicit biases in terms of which patients are thought to use substances, which types of substances they might use and even more critically, which type of treatment, if any, they are offered. Implicit biases combine with the effects of systemic racism to compound these consequences. It’s important to note that it’s not race that drives poor health outcomes, but racism.  W: Challenging stigma is one of the most powerful ways that detailing campaigns can combat the damage done by the War on Drugs, because stigma can make the difference of whether or not people receive dignified care. With a campaign so focused on addressing stigma and with a topic this important, how do you prepare your detailers for this task? C: We devote a significant amount of time towards training our detailing reps – a week-long training, 8 hours a day. We spend a large amount of that time talking in detail about stigma as related to cocaine use. It’s critical to us that our detailers are comfortable and knowledgeable when speaking about this topic, because it sets the tone for the providers who then set the tone for their patients. We ensure that our representatives are prepared to respond to a wide range of questions or comments, because this builds the provider-detailer relationship and enhances the value of the detailing visit. We’ve found during our follow-up visits that this support has led to high provider engagement with the campaign and providers reporting incorporation of the key recommendations into their daily practice, which is the aim of our public health detailing campaigns.  W: How have providers responded when detailed on a topic that carries so much stigma? C: The good news is that we’ve found NYC healthcare providers to not only be receptive to our work on substance use, but they’re eager to partner with us to support their patients once they learn about the severity of the issue. Our team provides statistics that relate to the provider’s specific neighborhoods and specialty, giving them real-time pictures of what’s happening with the patients they see. We know that it’s still a difficult topic to bring up, so we help address this with our action kit resources on stigmatic language and counter-top brochures that signal to patients that the provider’s office is a safe place to discuss these issues.  W: It gives me tremendous hope to hear about that there’s been enthusiastic response from providers. It means that things are changing. Let’s also talk a bit about program sustainability. Your team has worked extensively on campaigns across multiple topics. What have you learned from implementing past campaigns? C: Each public health detailing campaign is different, but we’ve learned some key strategies that support the growth and success of subsequent campaigns: 
Our overall goal is to do everything we possibly can to improve the health of our fellow New Yorkers. I like to remind our detailers of this James Baldwin quote that informs our public health detailing mission: “Not everything that is faced can be changed, but nothing can be changed until it is faced.” Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Carla Foster, MPH is an Epidemiologist at the New York City Department of Health and Mental Hygiene (NYC DOHMH). Her research focuses on the implementation and evaluation of public health detailing campaigns across New York City with the aim of reducing overdose mortality. Prior to joining the NYC DOHMH, she led development of clinical practice guidelines at the American Urological Association. She received dual Bachelor of Arts degrees in Africana Studies and Neuroscience from Wellesley College. Carla also obtained her Master of Public Health Degree in Epidemiology from Columbia University. A Team Effort: Strong Provider-Detailer Relationships to Amplify Evidence-Based Care (Part II)11/3/2020
Overview: The DETAILS blog presents a special two-part series of what it takes to build a strong provider-detailer relationship from the perspective of a long-time academic detailer and then, from one of her local physician partners that’s received AD for almost 15 years. You can read Part One here. In Part Two, we get a chance to speak with Dr. Robert ‘Bob’ Schwartz, a Vermont family physician and medical director who discusses the impact of AD on his clinical practice. Bob reflects on what a strong provider-detailer relationship looks like from the perspective of a clinician, especially in the midst of COVID-19, and offers his advice to other providers considering AD. An interview with Winnie Ho, NaRCAD Program Coordinator. Tags: Detailing Visit, Evidence-Based Medicine  Winnie: We’re so excited to have the chance to speak with a clinician who’s been receiving AD for a long time! Can you tell us a little bit about how you came to work with the Vermont Academic Detailing Program? Bob: It’s been several years that I’ve been blessed with Amanda Kennedy’s presence on the AD service. I would say that it actually all started back when we first decided we were not going to allow pharmaceutical representatives in the office about 20 years ago. We decided that it wasn’t a good use of our time. Then Amanda and the Vermont Academic Detailing Service came onto the scene. They presented a formal alternative to learn about medications from a non-biased view – and so we’ve participated in every single session since then. W: That’s incredible!  B: The program is great – I think what’s really critical is that the program has such a high level of professionalism that you can be completely trusting of the information that you’re getting. My colleagues and I are always so thrilled when there’s another topic ready. I know how long it takes to get these things set up. You know it’s not just something someone slaps together on a random afternoon. Amanda is an absolutely amazing professional, and the fact that she’s been with us for so long helps develop a personal relationship and it builds trust. W: This relationship has been developed over more than a decade! Here at DETAILS, we don’t always get a chance to ask about what happens after the detailer leaves the office. What challenges in clinical care make it critical to lean on Amanda as a trusted source of evidence and resources? B: I like that AD is not about a specific medication, but rather a specific medical condition. As a clinician, I think about the patient sitting in front of me with COPD, not about who I can get on a specific drug. Pharmaceutical detailers only talked about specific medications and you couldn’t trust the information as unbiased. They never gave you the whole picture. What’s really important in medicine is how all the pieces fit together, and that’s really hard as a clinician to figure out on your own. With AD, I can reach out to say that I have COPD patients and I really want to get more knowledge about COPD.  Dr. Schwartz's desk with AD Materials accumulated through the years from the Vermont Academic Detailing Program Dr. Schwartz's desk with AD Materials accumulated through the years from the Vermont Academic Detailing Program W: Right, and there’s an abundance of research that’s difficult to sort through on your own. B: AD is one of the few places that you can get this comprehensive evaluation of a specific condition and the medications that surround that. The other thing is that they bring materials. It might be a one-page sheet with diabetes medications based on class, relative cost, brand and generic names, and dosage. Or, it might be a COPD assessment score tool that I can easily refer to. These clinical aids are a big part of what detailers bring for us. W: Absolutely – those materials take a long time to create! I wanted to dive a little deeper into the provider-detailer relationship. What role do you think trust plays in sustaining a provider-detailing relationship for the long-term?  B: When we ask Amanda a question that she doesn’t know the answer to, she will tell us that she doesn’t know, but will look into it. A week later, you get an e-mail with information that she’s put together. We trust her and know that the dynamic of the relationship is not manipulative. We know that the information is carefully researched and that she’s not going to fill in the blanks by winging it. All of our detailing sessions have an unstructured portion where we can ask specific questions of Amanda. It allows the providers in our practice to have an organic conversation about a specific issue, and it can be hard to replicate this without the support of a detailing program. W: It’s always important to acknowledge what we know and what we don’t know, especially right now with COVID-19. It’s disrupted a lot of things, and I imagine for our healthcare providers, more than ever, there’s a lot of extra challenges. Our detailers’ main goals have always been to support their local providers and to be there with them through obstacles as they arise. What would you want detailers to understand about the challenges that COVID has brought on for clinicians?  B: Everything has been changing over the last several months and navigating COVID challenges takes up a lot of time. However, we still have to take care of our diabetes and our COPD patients and so more than ever, it’s critical that we have access to information that is streamlined, accurate, and that we can be confident in. I always tell people that medicine and life are team sports, and if you think you’re going to excel at either one of those alone, you’re going to be disappointed every time. We need Amanda on our team because she fills this vital role, that’s even more vital to have filled today. W: And finally, we know that there’s a lot of other clinicians who may be on the fence about receiving AD because they’re unfamiliar with it, or may have lingering distrust from pharmaceutical representatives. As someone who’s received AD for a long time, what advice would you give to other providers who may be considering these visits?  B: I conceptualize AD like this – it’s like getting on the super highway instead of taking the secondary roads. I could drive to Chicago and never leave a two-lane highway, or I can get on the 90 and drive on that to Chicago all the way. To other clinicians – we have certain responsibilities to ourselves and our patients, and one of those responsibilities is to keep up with what’s going on and to know how to use it to better care for our patients. What I would tell other clinicians is that, you have this responsibility anyway – why not have another team member who’s an expert on this and give this information to you in an hour what would have taken you six hours to do yourself? We’ve loved our partnership with Amanda, and we look forward to what comes next out of it. Have thoughts on our DETAILS Blog posts? You can head on over to our Discussion Forum to continue the conversation!  Bob Schwartz, MD, is a family physician at Northshire Medical Center in Manchester, Vermont. He’s served as the Associate Medical Director for Dartmouth Hitchcock Putnam, a multi-specialty group in the southwestern part of Vermont. Dr. Schwartz completed his family medicine training at Lancaster General Hospital in Lancaster, PA following his medical education in the Honors program at Northwestern University. He currently lives in East Dorset, Vermont. |
Highlighting Best PracticesWe highlight what's working in clinical education through interviews, features, event recaps, and guest blogs, offering clinical educators the chance to share successes and lessons learned from around the country & beyond. Search Archives
|

